 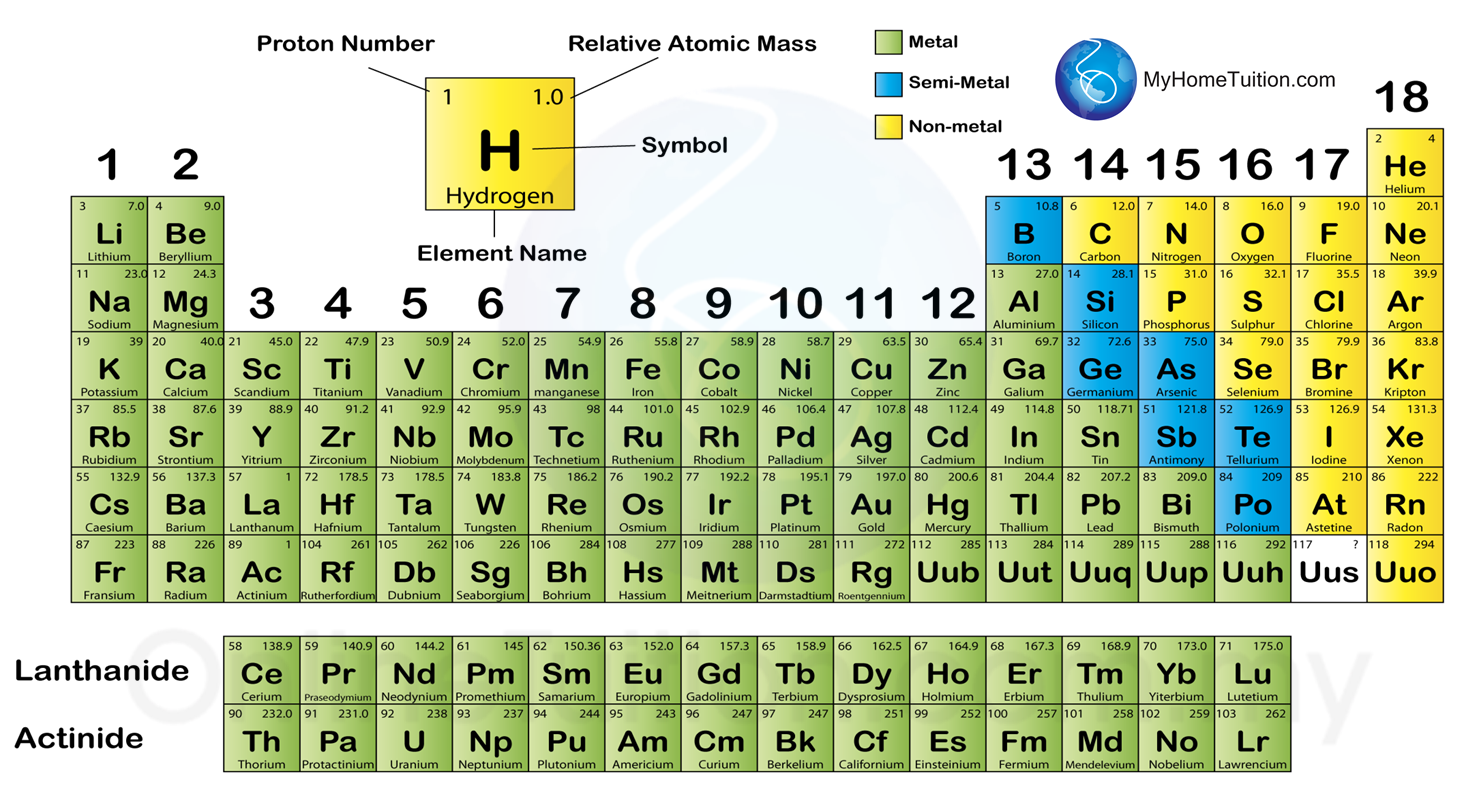
The new IUPAC scheme was developed to replace both systems as they confusingly used the same names to mean different things. This section will seek to explore some of the wider patterns of reactivity. this is because they share common patterns in the number of electrons and shells but differ by the number of protons and neutrons. The former system was frequently used in Europe while the latter was most common in America. Most groups in the periodic table have common patterns in reactivity. In the old IUPAC system the letters A and B were designated to the left (A) and right (B) part of the table, while in the CAS system the letters A and B were designated to main group elements (A) and transition elements (B). There is considerable confusion surrounding the two old systems in use (old IUPAC and CAS) that combined the use of Roman numerals with letters. The Roman numeral names are the original traditional names of the groups the Arabic numeral names are those recommended by the International Union of Pure and Applied Chemistry (IUPAC) to replace the old names in an attempt to reduce the confusion generated by the two older, but mutually confusing, schemes. What Are Periods & Groups In The Periodic Table Properties of Matter Chemistry FuseSchoolWhat's the difference between periods and. There are three ways of numbering the groups of the periodic table, one using Hindu-Arabic numerals and the other two using Roman numerals. In chemistry, a group (also known as a family)1 is a column of elements in the periodic table of the chemical elements. The modern explanation of the pattern of the periodic table is that the elements in a group have similar configurations of the outermost electron shells of their atoms: as most chemical properties are dominated by the orbital location of the outermost electron. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.What is the Correct Way to Check Repeatability in Balances? Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Free Gift for you: Interactive Periodic Table In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). Group 17 elements are highly reactive and non-ferrous metals. 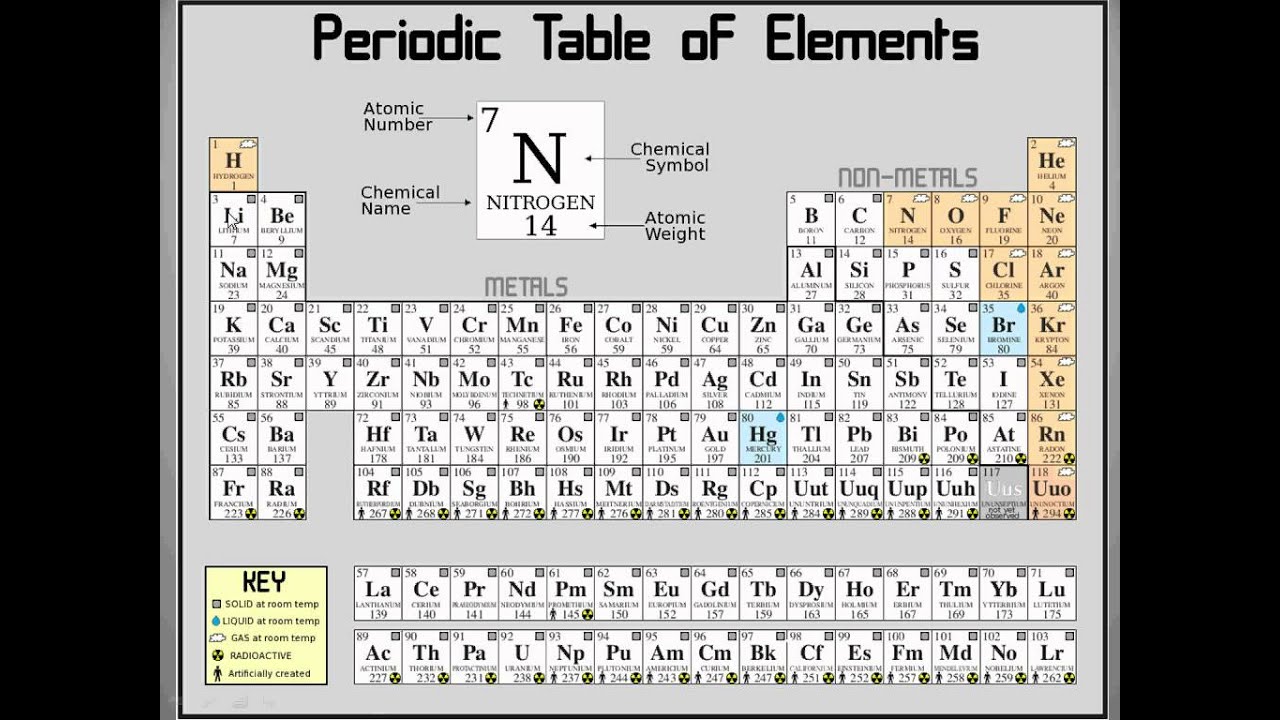 /the-periodic-table--digital-illustration--73016803-598b218ec41244001024af78.jpg)
For example, Group 1 elements are all soft and reactive metals. Elements within the same group have the same number of valence. The elements of a group have chemical and physical properties in common. Metals are found on the left of the periodic table, whilst non-metals are found on the. The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. The group/family number is the number assigned to the vertical columns of the periodic table. You can also classify elements as metals, non-metals, and metalloids. There are total 18 vertical columns on periodic table. From left to right in the periodic table, there are two groups (1 and 2) of elements in the s-block, or hydrogen block, of the periodic table ten groups (3 through 12) in the d-block, or transition block and six groups (13 through 18) in the p-block, or main block. Groups are the vertical columns on the periodic table. 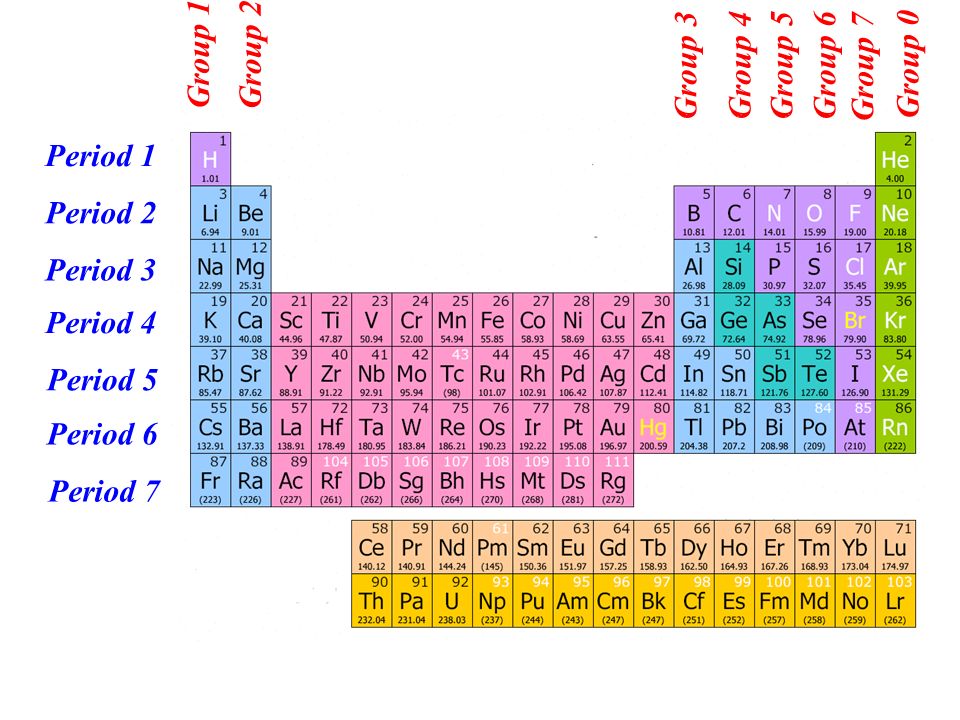
In terms of electronic structure of the atom, a group constitutes a series of elements. The elements included in Noble gases group are įor detailed information on noble gases, read the main article on Noble gases of periodic table. Elements that have similar chemical properties are grouped in columns called groups (or families) A column of elements on the periodic table. A group may be defined as vertical column in the periodic table. Noble gases group is the last group (group 18) on the periodic table. There are multiple ways of grouping the elements, but they are commonly divided into metals, semimetals (metalloids), and nonmetals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
